Abstract: S151
Type: Presidential Symposium
Presentation during EHA23: On Friday, June 15, 2018 from 16:15 - 16:30
Location: Room A1
Background
Obinutuzumab (GA101; G), a glycoengineered type
II anti-CD20 monoclonal antibody, has been developed as an effective
treatment for chronic lymphocytic leukemia (CLL). The phase III CLL11
study evaluated the efficacy and safety of G plus chlorambucil (Clb;
G-Clb) and rituximab (R) plus Clb (R-Clb) vs Clb alone (Stage 1), as
well as G-Clb vs R-Clb (Stage 2), in patients with previously untreated
CLL and comorbidities. Previous pre-planned analyses have established
superiority of G-Clb over Clb alone and R-Clb.
Aims
We report the final analysis of the CLL11 study (data
cut-off, 10 October 2017), with approximately 2 years of additional
follow-up compared with previous analyses.
Methods
Patients were randomised 1:2:2 to receive six
28-day cycles (C) of Clb, R-Clb or G-Clb. Clb (0.5 mg/kg) was
administered orally on Day (D) 1 and D15 of C1–6. R was administered
intravenously (IV) at a dose of 375 mg/m2 on D1 of C1 and 500 mg/m2 on
D1 of C2–6. G (1000 mg) was administered IV on D1 (dose split over two
days; 100 mg D1, 900 mg D2), D8 and D15 of C1, and D1 of C2–6. Eligible
patients had previously untreated CD20+ CLL, a total Cumulative Illness
Rating Scale (CIRS) score of >6 and/or a creatinine clearance (CrCl)
of <70 mL/min. The primary endpoint was investigator-assessed
progression-free survival (PFS). Secondary endpoints included overall
survival (OS), time to new treatment (TTNT) and safety. Comparison of
PFS was performed by log rank test. Treatment effects were expressed as
hazard ratios (HR) using a stratified Cox regression model.
Results
A total of 781 patients were enrolled and received
treatment (median: age, 73 years; CIRS score, 8; CrCl, 62 mL/min). No
new safety signals were identified at this update. After a median
observation time of 62.5 months, treatment with G-Clb (n=238) was
associated with improved outcomes compared with Clb alone (n=118);
median: PFS, 31.1 vs 11.1 months (HR 0.21, 95% CI 0.16–0.28,
p<0.0001); OS, not reached vs 66.7 months (HR 0.68, 95% CI 0.49–0.94,
p=0.0196); and TTNT, 55.7 vs 15.1 months (HR 0.25, 95% CI 0.19–0.35,
p<0.0001). After a median observation time of 59.4 months, G-Clb
(n=333) also demonstrated a clinically meaningful improvement in
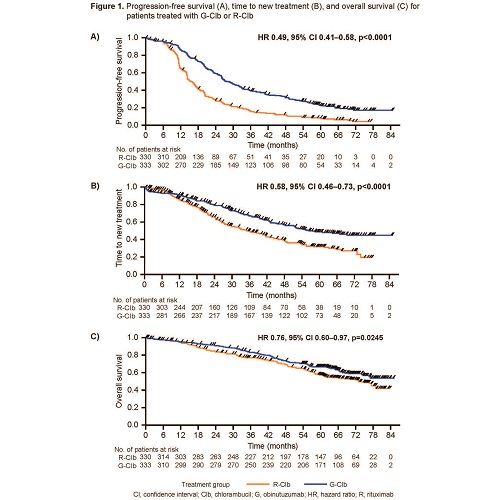
outcomes compared with R-Clb (n=330); median: PFS, 28.9 vs 15.7 months
(HR 0.49, 95% CI 0.41–0.58, p<0.0001) and TTNT, 56.4 vs 34.9 months
(HR 0.58, 95% CI 0.46–0.73, p<0.0001) (Figure 1A and 1B).
Notably, G-Clb also provided a clinically meaningful improvement in OS
compared with R-Clb; median OS, not reached vs 73.1 months (HR 0.76, 95%
CI 0.60–0.97, p=0.0245) (Figure 1C). Two- and
five-year survival rates were 91% vs 84% and 66% vs 57% for G-Clb vs
R-Clb, respectively. Overall, fewer patients died in the G-Clb arm (37%)
than in the R-Clb arm (45%). During the survival follow-up period, the
most common cause of death was disease progression (G-Clb, 10%; R-Clb,
15%).
Conclusion
This final survival analysis from the CLL11
study confirms that G-Clb provides clinically meaningful benefits in CLL
patients with comorbidities, including prolongation of PFS and OS, when
compared with R-Clb and Clb alone, and an absolute treatment-free
duration of approximately four and a half years, while maintaining an
acceptable and manageable safety profile. These findings support the use
of G-Clb as first-line treatment for CLL patients with comorbidities,
and suggest G as the preferred anti-CD20 antibody in future combination
regimens for CLL.
Session topic: 6. Chronic lymphocytic leukemia and related disorders - Clinical






















